- Home
-
-
-
Stiles Enhancement Technologies
-
-
-
-
- Stiles can introduce you to advances in software integration. From raw materials to design, from production to shipping, our solutions help you manage your manufacturing processes by delivering more powerful information and greater control.
- Integration
- Data Development
- Training
- Maintenance & Support
-
-
-
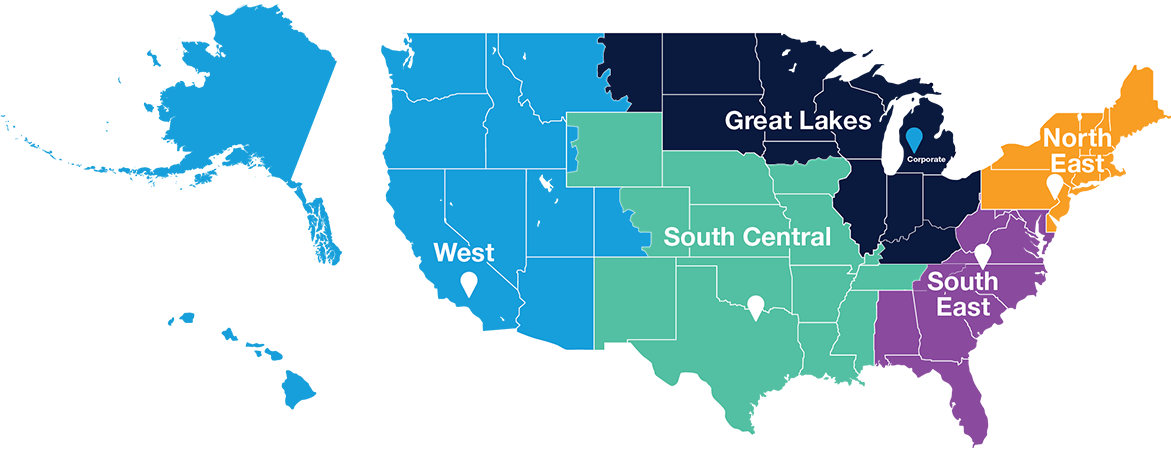
- Stiles Machinery hosts a national network of five regional service centers and nationwide force of over 200 Field Service Representatives put the industry’s best service where it belongs – close to you.
- Routine & Preventative Maintenance
- Technical Support & Diagnostics
- Field Service
- Request an Equipment Manual
-
For support and service of your machinery, Stiles has experts available 24/7 to help reduce downtime and get you back in action.
Technical Support: 616-698-6615
Service & Repairs: 616-698-7500
24/7 Parts: 1-800-727-8780
sale@inlandmachineokc.com
-
-
Parts are available from Stiles 24 hours a day, seven days a week, and are shipped from our Grand Rapids, Michigan fulfillment center.
24/7 Parts: 800-727-8780
-
-

With a customized Stiles University course, we can provide expert training for your team, on-location. Whether it's programming, operation or maintenance, we want to help you train your personnel to meet your production goals efficiently and effectively.
-

Experience the trusted workforce development training and machinery knowledge you rely on from Stiles University, now at your convenience. With technology changing every day and new methods being developed constantly, Stiles University Online has the latest information and training content available at your fingertips.
-
Your solution for workforce development.
616-698-7500
-
-
-

Do you want to produce more efficiently? Processes and flows are key. We optimize these together with you, re-organize them and make sure that you reduce your lead time and save costs. This enables you to implement customized manufacturing and achieve your business goals.
-

Industrialized construction is evolving. Automation, robotics and advanced technology are raising the level of productivity, efficiency and precision for builders in North America.
-

Stiles Machinery is at the forefront of providing technology and machining for producing high quality mass timber. Automated solutions for your mass timber production can increase your manufacturing quality and productivity.
-

Project management services from Stiles make it easy to streamline your entire project— from concept and consultation to integration and implementation.
-
- Online Store
- News
- Careers
- REQUEST INFO
- Home
-
-
-
Stiles Enhancement Technologies
-
-
-
-
- Stiles can introduce you to advances in software integration. From raw materials to design, from production to shipping, our solutions help you manage your manufacturing processes by delivering more powerful information and greater control.
- Integration
- Data Development
- Training
- Maintenance & Support
-
-
-
- Stiles Machinery hosts a national network of five regional service centers and nationwide force of over 200 Field Service Representatives put the industry’s best service where it belongs – close to you.
- Routine & Preventative Maintenance
- Technical Support & Diagnostics
- Field Service
- Request an Equipment Manual
-
For support and service of your machinery, Stiles has experts available 24/7 to help reduce downtime and get you back in action.
Technical Support: 616-698-6615
Service & Repairs: 616-698-7500
24/7 Parts: 1-800-727-8780
sale@inlandmachineokc.com
-
-
Parts are available from Stiles 24 hours a day, seven days a week, and are shipped from our Grand Rapids, Michigan fulfillment center.
24/7 Parts: 800-727-8780
-
-

With a customized Stiles University course, we can provide expert training for your team, on-location. Whether it's programming, operation or maintenance, we want to help you train your personnel to meet your production goals efficiently and effectively.
-

Experience the trusted workforce development training and machinery knowledge you rely on from Stiles University, now at your convenience. With technology changing every day and new methods being developed constantly, Stiles University Online has the latest information and training content available at your fingertips.
-
Your solution for workforce development.
616-698-7500
-
-
-

Do you want to produce more efficiently? Processes and flows are key. We optimize these together with you, re-organize them and make sure that you reduce your lead time and save costs. This enables you to implement customized manufacturing and achieve your business goals.
-

Industrialized construction is evolving. Automation, robotics and advanced technology are raising the level of productivity, efficiency and precision for builders in North America.
-

Stiles Machinery is at the forefront of providing technology and machining for producing high quality mass timber. Automated solutions for your mass timber production can increase your manufacturing quality and productivity.
-

Project management services from Stiles make it easy to streamline your entire project— from concept and consultation to integration and implementation.
-
- Online Store
- News
- Careers
- REQUEST INFO
- Home
-
-
-
- Stiles can introduce you to advances in software integration. From raw materials to design, from production to shipping, our solutions help you manage your manufacturing processes by delivering more powerful information and greater control.
- Integration
- Data Development
- Training
- Maintenance & Support
-
- TECHNICAL SUPPORT
- FIELD SERVICE
- ROUTINE & PREVENTATIVE MAINTENANCE
- REQUEST AN EQUIPMENT MANUAL
-
For support and service of your machinery, Stiles has experts available 24/7 to help reduce downtime and get you back in action.
Technical Support: 616-698-6615 Service & Repairs: 616-698-7500 24/7 Parts: 1-800-727-8780
-
-
Parts are available from Stiles 24 hours a day, seven days a week, and are shipped from our Grand Rapids, Michigan fulfillment center.
24/7 Parts: 800-727-8780
-
- IN-PERSON TRAINING
- CUSTOMIZED COURSES & TRAINING
- STILES UNIVERSITY ONLINE
-
Your solution for workforce development.
- Online Store
- News
- Careers
- FINANCING
- Blog
- REQUEST INFO
- Home
-
-
-
- Stiles can introduce you to advances in software integration. From raw materials to design, from production to shipping, our solutions help you manage your manufacturing processes by delivering more powerful information and greater control.
- Integration
- Data Development
- Training
- Maintenance & Support
-
- TECHNICAL SUPPORT
- FIELD SERVICE
- ROUTINE & PREVENTATIVE MAINTENANCE
- REQUEST AN EQUIPMENT MANUAL
-
For support and service of your machinery, Stiles has experts available 24/7 to help reduce downtime and get you back in action.
Technical Support: 616-698-6615 Service & Repairs: 616-698-7500 24/7 Parts: 1-800-727-8780
-
-
Parts are available from Stiles 24 hours a day, seven days a week, and are shipped from our Grand Rapids, Michigan fulfillment center.
24/7 Parts: 800-727-8780
-
- IN-PERSON TRAINING
- CUSTOMIZED COURSES & TRAINING
- STILES UNIVERSITY ONLINE
-
Your solution for workforce development.
- Online Store
- News
- Careers
- FINANCING
- Blog
- REQUEST INFO
What is a strong base anion resin and how does it work?
strong base anion resin is a vital component in water treatment processes. This resin plays a significant role in removing unwanted anions from water. Its structure allows for efficient ion exchange, attracting negatively charged ions.
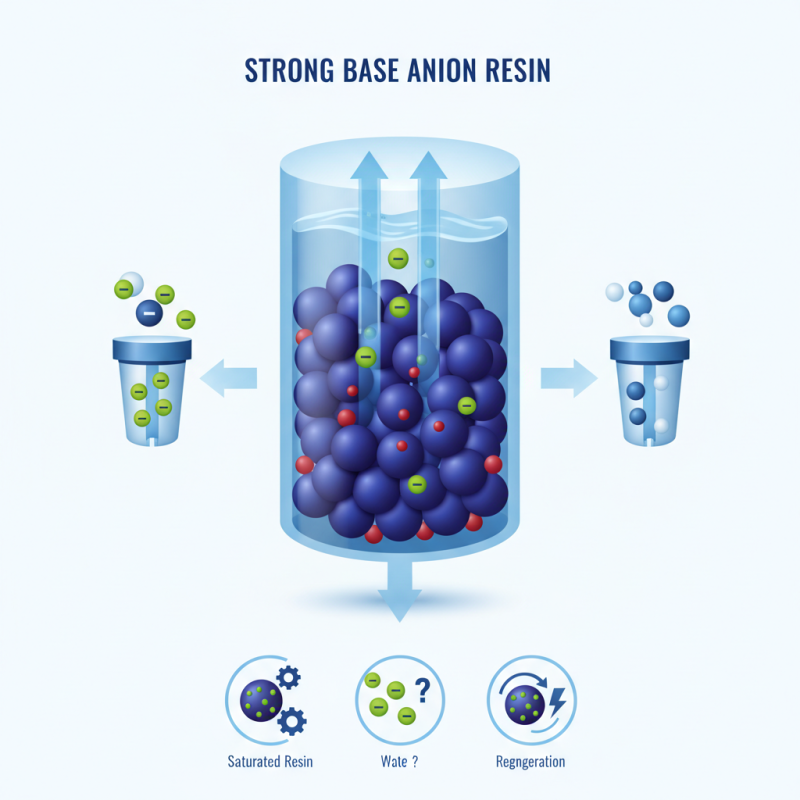
The mechanism of strong base anion resin involves interaction with these harmful ions. When water flows through the resin, the resin grains exchange chloride ions for the anions present in the water. This process reduces contaminants effectively. The result is cleaner, safer water for various applications.
However, some challenges exist in using strong base anion resin. Resin saturation occurs over time, requiring regeneration to maintain efficiency. Additionally, not all anions are captured equally. Users must consider these factors when implementing this technology. This reflection can lead to better water treatment strategies.
Definition of Strong Base Anion Resins
Strong base anion resins are crucial in various industries, particularly in water treatment and chemical processing. These resins are designed to exchange hydroxide ions with anions from solutions. Their effectiveness stems from their unique structure, often composed of a polystyrene backbone with quaternary ammonium functional groups. They operate efficiently in neutral to alkaline pH ranges, capturing contaminants while allowing for water purification.
Recent industry reports indicate that the global demand for anion exchange resins, including strong base types, has been on the rise. Analysts predict a growth rate of over 6% annually through 2025. This increase is linked to the need for clean water sources and efficient waste management solutions. Strong base anion resins excel at removing nitrates, sulfates, and chlorides, making them valuable in municipal water systems.
However, it's essential to consider the limitations of these resins. Regeneration processes can be complex and require specific conditions. If not managed properly, they can lead to resin degradation. The balance between performance and maintenance needs often raises concerns among operators. Understanding these aspects is vital for achieving optimal use of strong base anion resins in practical applications.
Composition and Structure of Strong Base Anion Resins
Strong base anion resins are essential in water treatment processes. Their structure typically consists of a polymer matrix containing quaternary ammonium groups. This design allows them to capture and exchange anions effectively. They are commonly used to remove contaminants like nitrates and sulfates from water.
The composition of these resins usually features cross-linked polystyrene. The cross-linking enhances stability and durability. The quaternary ammonium functional groups attract various negatively charged ions. As they pass through the resin, undesirable anions are exchanged for hydroxide ions, purifying the water.
Tip: When working with strong base anion resins, ensure proper pH balance. This can optimize their performance in specific applications.
These resins can degrade under extreme conditions. Factors like high temperature and concentration can reduce their lifespan. It's crucial to monitor their performance regularly.
Tip: Consider conducting routine tests to determine resin efficiency. Early detection of issues leads to better maintenance and longer service life.
Mechanism of Ion Exchange in Strong Base Anion Resins
Strong base anion resins are essential in water treatment processes. They work by exchanging negatively charged ions. In this mechanism, anion resins capture unwanted contaminants and release hydroxide ions. This process enhances water purity significantly.
During ion exchange, the resin's functional groups attract anions from water. For instance, chloride ions can be replaced by sulfate ions. The resin holds onto these contaminants. Reports indicate that strong base anion resins can remove over 95% of specific anions. This efficiency makes them critical in various industries.
Tip: Ensure the resin is regenerated regularly. Degraded resin performance may lead to poor water quality.
An important factor is the pH level. The effectiveness of ion exchange can diminish at extreme pH. Regular monitoring is essential to maintain optimal performance. Operating conditions must be controlled to ensure maximum efficiency.
Tip: Always check for scaling or fouling. These issues can reduce the effectiveness of the anion exchange process. Potential loss in performance needs to be addressed proactively.
Applications of Strong Base Anion Resins in Water Treatment
Strong base anion resins are essential in water treatment. They work by exchanging hydroxide ions for anions in the water. This process removes contaminants like nitrates and sulfates, ensuring water quality. The efficiency of these resins can significantly impact purification levels.
In industrial settings, the demand for clean water is growing. A recent report states that 60% of wastewater treatment plants in urban areas use anion resins. These plants achieve a reduction of over 90% in harmful anions. Such effectiveness is vital for maintaining environmental standards. However, improper resin management can lead to reduced performance and increased costs.
These resins also have diverse applications beyond water treatment. They are used in food and beverage purification. Industries are noticing that strong base anion resins can lower harmful substances in products. Nonetheless, there’s a gap in optimizing their lifecycle. Companies must focus on better maintenance protocols. Ignoring this can lead to subpar water quality, which raises long-term concerns.
Advantages and Limitations of Strong Base Anion Resins
Strong base anion resins are widely used in water treatment and purification processes. They have unique properties that allow them to effectively remove negatively charged ions. These resins are often utilized in deionization systems. They excel in filtering out harmful contaminants. However, their effectiveness can vary depending on the specific application.
One of the key advantages of strong base anion resins is their high capacity for ion exchange. They can handle varying pH levels efficiently. This makes them suitable for diverse industrial applications. Their durability is another notable benefit. These resins can last a long time if properly maintained. Nevertheless, they can also be vulnerable to fouling. This may limit their use in certain conditions, requiring frequent monitoring.
Another limitation is the potential for chemical incompatibility. Some substances can degrade the resin's structure. This leads to reduced efficiency over time. Additionally, the initial cost of these resins can be higher than other options. Organizations must weigh the benefits against the expenses. In certain scenarios, this technology may not be the best fit. Users should consider all factors before making decisions.
What is a strong base anion resin and how does it work? - Advantages and Limitations of Strong Base Anion Resins
| Property | Details |
|---|---|
| Definition | A type of resin that can exchange anions from solutions, typically used in water treatment and purification processes. |
| Function | Strong base anion resins operate by exchanging hydroxide ions (OH⁻) in exchange for anions in water. |
| Common Applications | Water deionization, wastewater treatment, and in various industrial processes. |
| Advantages | High selectivity for anions, effective in removing contaminants, can regenerate easily. |
| Limitations | Higher cost compared to weak base resins, can be sensitive to temperature and pH levels. |
| Regeneration | Regenerates using sodium hydroxide (NaOH) or other suitable hydroxide solutions. |
| Lifespan | Resins can last several years depending on usage and maintenance practices. |